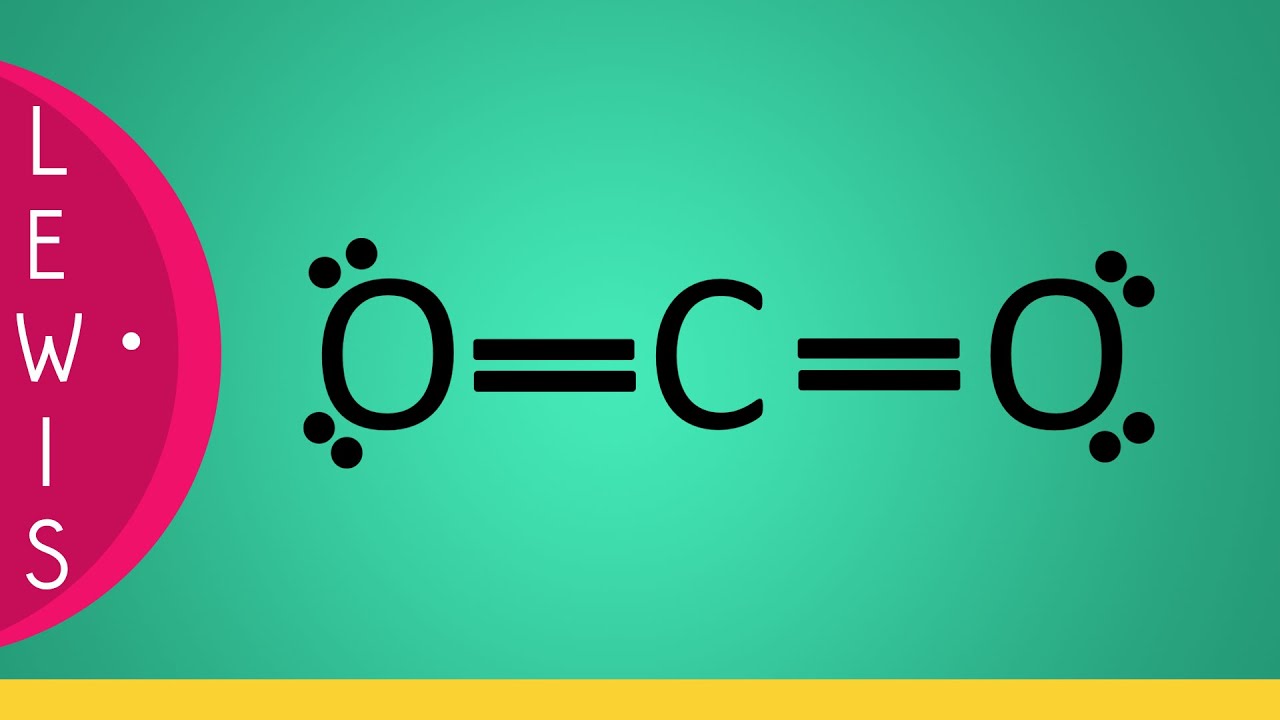
CO2 Lewis Structure YouTube
1: Basic Concepts in Chemical Bonding and Organic Molecules
Carbon Dioxide Electron Dot Diagram General Wiring Diagram
A step-by-step explanation of how to draw the Lewis Dot Structure for Carbon dioxide (CO2 ).For the Carbon dioxide structure we use the periodic table to fi.

CO2 Lewis Structure ,Valence Electrons, Formal Charge ,Polar or
The lewis structure of CO2 can be with some simple steps, but before that, it is important to understand lewis structure properly. So lewis structure generally gives us an idea about the nature of bonding and octet fulfillment of the atoms. According to the octet rule, an atom attains stability by fulfilling its octet.
Molecular Geometry, Lewis Structure, and Bond Angle of CO2
665 109K views 3 years ago This chemistry video explains how to draw the lewis structure of CO2 also known as Carbon Dioxide. It also discusses the bond angle, molecular geometry, and.

Lewis Structure Carbon Dioxide Co2 Stock Vector (Royalty Free
A step-by-step explanation of how to draw the CO2 Lewis Dot Structure (Carbon dioxide).For the CO2 structure use the periodic table to find the total number.

CO2 Lewis Structure, Hybridization, Molecular Geometry, and MO Diagram
Lewis structure of CO2 (or Carbon Dioxide) contains two double bonds between the Carbon (C) atom and each Oxygen (O) atom. The Carbon atom (C) is at the center and it is surrounded by 2 Oxygen atoms (O). The Carbon atom does not have a lone pair while both the Oxygen atoms have 2 lone pairs.
[Solved] 8. Draw the Lewis structures for CO2 and CO, and predict the
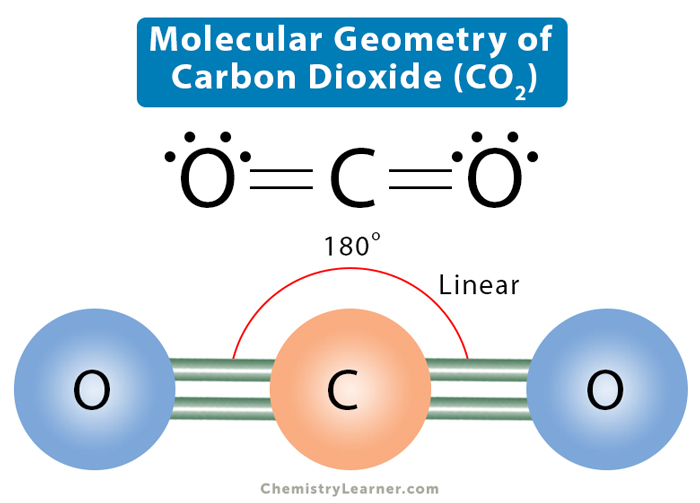
The Lewis structure helps us identify the bond pairs and the lone pairs. Then, with the Lewis structure, we apply the valence-shell electron-pair repulsion (VSPER) theory to determine the molecular geometry and the electron-group geometry.. Carbon dioxide has two electron groups and no lone pairs. Carbon dioxide is therefore linear in.

Molecular Structure of CO2 (Carbon Dioxide) YouTube
The CO2 Lewis structure depicts the molecular arrangement of carbon dioxide, which is composed of one carbon atom and two oxygen atoms. Within the CO 2 Lewis structure, the carbon atom is surrounded by two double bonds, with each oxygen atom attached to it. Additionally, there are two lone pairs on each oxygen atom.

CO2 Molecular Geometry Science Education and Tutorials
Lewis Structure Co2 Lewis Structure of Carbon Dioxide Carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. The carbon-oxygen ratio in a CO 2 molecule is 1:2. Two double bonds connect the carbon and oxygen atoms in the Lewis structure.
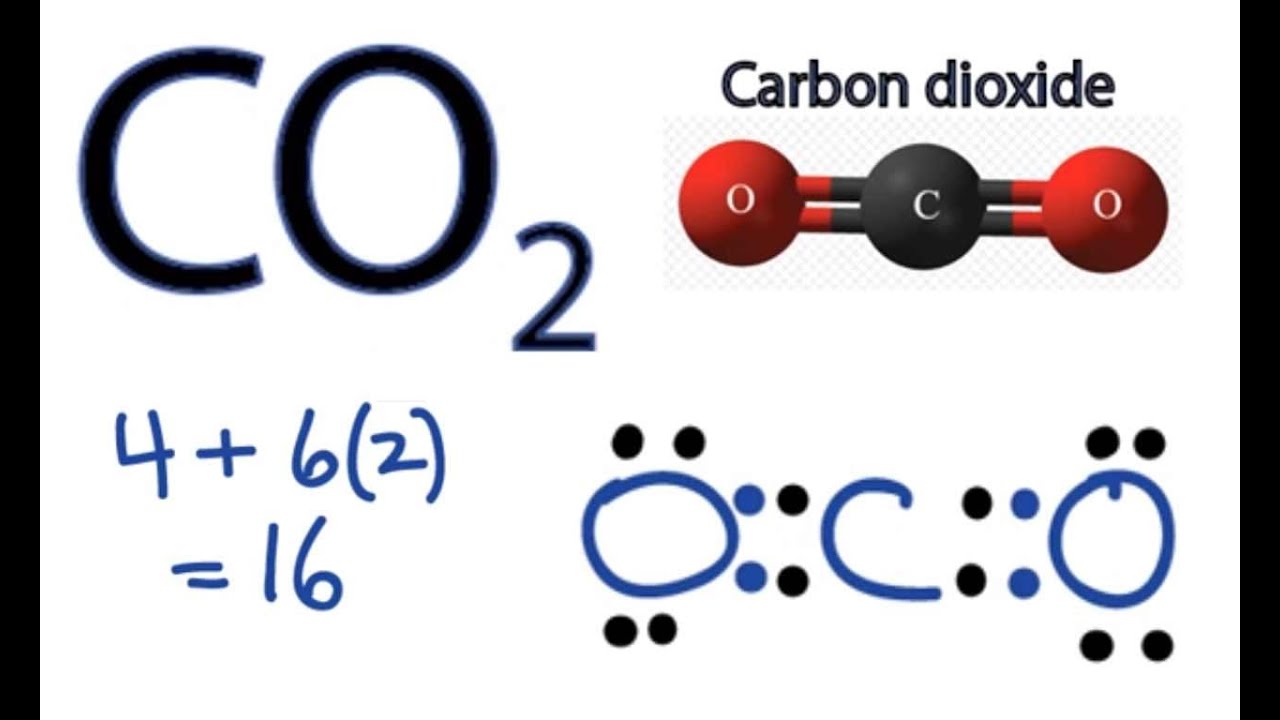
CO2 Lewis Structure How to Draw the Dot Structure for Carbon Dioxide
The Lewis dot structure of carbon dioxide is shown in Figure \(\PageIndex{8}\). Figure \(\PageIndex{8}\): Three resonance structures of carbon dioxide. In the above Figure we see the second and third resonance structures average out to the first, and so the average of all the resonance structures is a double bond. Thus it is common to write.

The WikiPremed MCAT Course Image Archive A lewis structure showing
The Lewis structure indicates that each Cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). A dash (or line) is sometimes used to indicate a shared pair of electrons: A single shared pair of electrons is called a single bond.

CO2 Lewis Structure ,Valence Electrons, Formal Charge ,Polar or
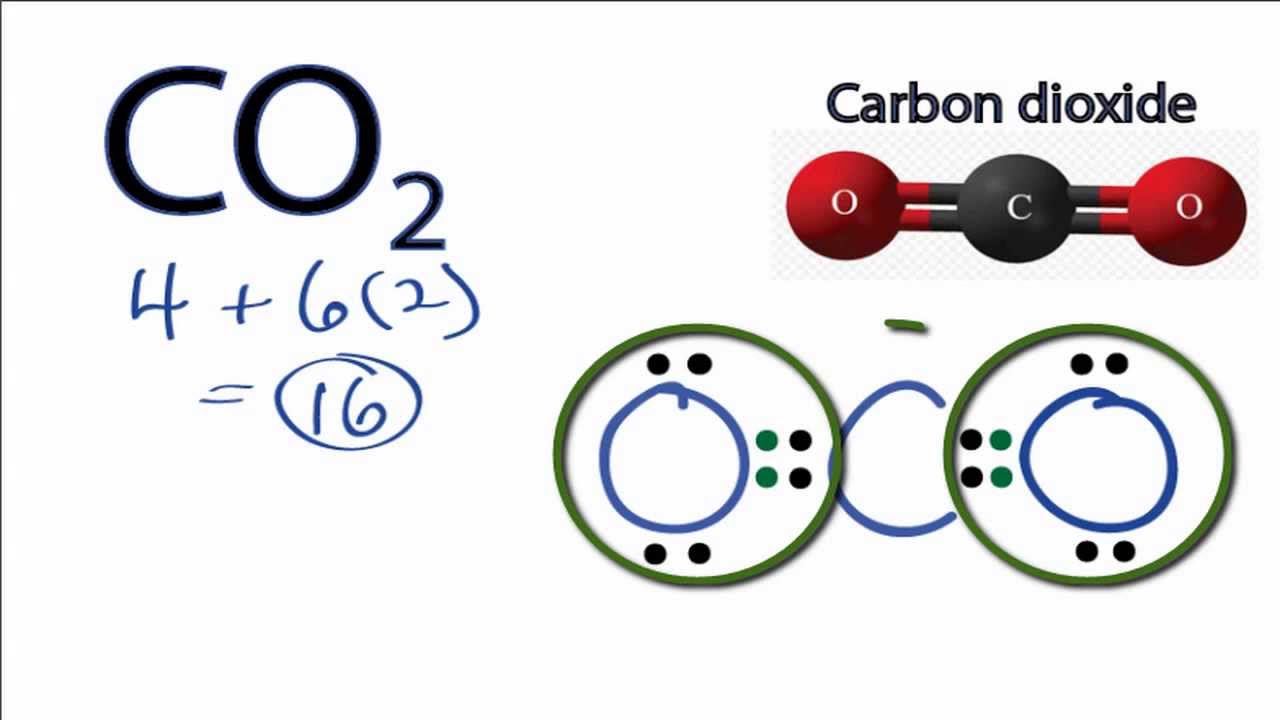
To draw the Lewis structure of CO2, we first need to determine the number of valence electrons in each atom. Carbon has 4 valence electrons, while each oxygen atom has 6 valence electrons. In total, CO2 has 16 valence electrons. Next, we need to arrange the atoms in the molecule.

What is the Lewis Dot structure for CO2 (Carbon dioxide)?
Lewis Dot Structure of CO2 (Carbon DiOxide) kentchemistry.com 25.1K subscribers Subscribe Subscribed 494 225K views 12 years ago Every Video I quickly take you through how to draw the.

What is the Lewis Dot structure for CO2 (Carbon dioxide)?
Steps of drawing CO2 lewis structure Step 1: Find the total valence electrons in CO2 molecule. In order to find the total valence electrons in CO2 (carbon dioxide) molecule, first of all you should know the valence electrons present in carbon atom as well as oxygen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)

What is the Lewis Dot structure for CO2 (Carbon dioxide)?
The CO 2 Lewis structure is symmetric. Generally, small symmetric molecules are nonpolar. CO 2 is a nonpolar substance, meaning it tends to be a gas. CO 2 has a rather low boiling point of around -80 ℃ or -100 ℉. It can be liquified and even frozen solid with special machinery to produce "dry ice.". Dry ice exhibits a temperature around.

So far, we’ve used 16 of the CO2 Lewis structure’s total 16 outermost
Carbon dioxide (CO 2) lewis structure has two oxygen atoms and one carbon atom. There are two double bonds around carbon atom in the CO 2. No lone pairs on carbon atom and each oxygen atom has two lone pairs on their valence shells. Shape of CO 2 is linear. Steps of drawing the lewis structure of CO 2 are explained in detail in this tutorial.